NEWS
We're here to support families of Duchenne muscular dystrophy.

🧬 What Happened FDA Request: On July 18, the FDA informally asked Sarepta to voluntarily halt all U.S. shipments of its Duchenne DMD gene therapy, Elevidys—a move driven by safety concerns after three patient deaths due to acute liver failure. This included two fatalities in teenage Duchenne patients treated with Elevidys and a third in an adult patient from a different Sarepta gene therapy trial . Initial Pushback: Sarepta initially resisted, stating there were no new safety concerns for ambulatory patients (those who can walk), and maintained shipments for that group. They had previously paused shipments for non-ambulatory patients earlier in the year after two deaths . Reversal and Pause: On July 21–22, Sarepta reversed course and voluntarily paused all shipments in the U.S. by end of business Tuesday, July 22, 2025, to cooperate with the FDA, respond to inquiries, and complete a revised safety labeling for Elevidys . --- 📌 Why It Matters Safety Labeling Update: The pause allows Sarepta and the FDA time to add a stricter warning (including a new black-box liver-risk warning) and possibly implement a Risk Evaluation and Mitigation Strategy (REMS) for monitoring liver safety . Regulatory Relations: CEO Doug Ingram labeled the decision “painful,” but necessary for maintaining a collaborative and constructive relationship with the FDA . Market Impact: Sarepta’s stock plunged, falling as much as 8–5% on the news, and is down nearly 90% year to date. Analysts warn that the pause and black-box warning could severely affect Elevidys uptake and 50–61% of Sarepta's revenue . --- 🔍 What’s Next Duration of Pause: The pause could extend 3–6 months while safety data is reviewed and label changes are finalized . Potential Outcomes: Resumption: Elevidys could return under new safety protocols/label. A REMS program may be required . Market Withdrawal: Analysts caution on the possibility of permanent withdrawal if FDA's safety concerns aren't resolved . Pipeline Impact: FDA has also placed a clinical hold on Sarepta’s other gene therapy trials (including for Limb-Girdle Muscular Dystrophy) and revoked platform tech designation due to the shared AAVrh74 viral vector . --- ✅ In Summary Key Takeaway Details Pause in effect All U.S. Elevidys shipments halted since end of business July 22 Why Safety concerns after 3 liver-related deaths from same viral vector Next steps Label update, FDA review, possible REMS; uncertain pause duration Risks Potential permanent withdrawal, impact on revenue, and pipeline delays

Aluminum is the third most abundant element in the Earth's crust and has a wide range of industrial and household applications. However, increasing evidence suggests that aluminum exposure may pose health risks, particularly concerning brain health and toxicity. In this article, we’ll explore what aluminum is, how it affects our bodies—especially the brain—and how it may contribute to neurodegenerative diseases like Alzheimer’s. We’ll also discuss sources of aluminum exposure, methods of testing for toxicity, and natural approaches to detoxification.

We are incredibly honored to share that JAR of Hope has been named the 2025 Non-Profit of the Year by the Monmouth Regional Chamber of Commerce as part of their prestigious Beacon Awards . The Beacon Awards are designed to recognize organizations that exemplify outstanding community enrichment through service, leadership, and achievement. It means so much to us that both the MRCC membership and the community at large recognized the work we do and the mission we live by every day. This award is not just a recognition of our team’s efforts—it’s a reflection of the amazing community that supports us, believes in our cause, and stands with us in the fight to raise awareness, fund research, and bring hope to families affected by Duchenne Muscular Dystrophy.
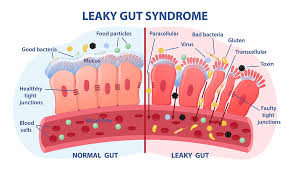
As research into autism spectrum disorder (ASD) continues to evolve, one intriguing area of study is the role of the microbiome—the trillions of microorganisms living in our guts. Emerging evidence suggests that the health of our microbiome may have significant implications for children with autism, particularly in relation to gut disorders and neurological development. This blog explores the connection between the microbiome and autism, emphasizing the importance of detailed stool analyses, checking for parasites, and understanding the gut-brain relationship. What is the Microbiome? The microbiome refers to the vast community of bacteria, viruses, fungi, and other microorganisms that inhabit our digestive systems. These microorganisms play a crucial role in digestion, immunity, and even brain function. In children with autism, studies have indicated that there may be an imbalance in the microbiome, often referred to as dysbiosis. This condition can lead to gastrointestinal issues that are commonly reported in children with ASD, such as constipation, diarrhea, and abdominal pain.

A groundbreaking study published in Cell on January 23, 2020, has identified 102 genes associated with autism spectrum disorder (ASD), including 30 previously unlinked to the condition. Led by Joseph Buxbaum, director of the Seaver Autism Center for Research and Treatment at Mount Sinai, the research analyzed over 35,000 DNA samples, nearly 12,000 of which were from individuals with autism. The study leveraged advanced genetic sequencing technologies and data from the Autism Sequencing Consortium, an international collaboration of scientists sharing samples and data since 2010. By comparing genetic sequences from individuals with autism to their siblings and to unaffected individuals, the researchers identified both inherited mutations and spontaneous (de novo) genetic alterations that may contribute to ASD. Notably, the research distinguished genes more closely associated with autism from those linked to other neurodevelopmental disorders, such as intellectual and motor disabilities. This differentiation enhances our understanding of autism's molecular mechanisms and could inform the development of targeted treatments. Dr. Geraldine Dawson, director of the Center for Autism and Brain Development at Duke University, hailed the study as "one of the most important, comprehensive studies on the genetics of autism to date," emphasizing its role in elucidating the complex genetic landscape of ASD. This research represents a significant step forward in autism genetics, offering hope for more personalized and effective interventions in the future. January 24, 2020 2:19 PM EST Time Magazine






